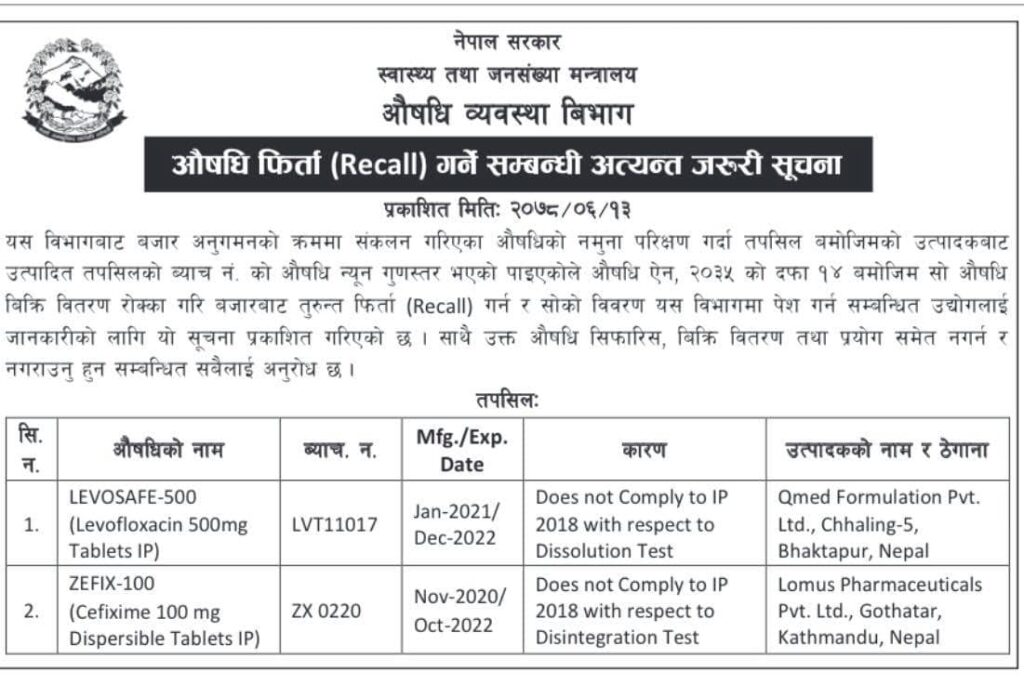
Department of Drug Administration Drug Recall Notice
While testing the samples of drugs collected during market monitoring from Department of Drug Administration – DDA, batch of details produced by the manufacturer as per the details no. The medicine was found to be of low quality.This information has been published for the information of the concerned industry and the official importer of the industry concerned and their representatives to immediately recall the drug from the market and submit its details to Drug Administration – DDA, by stopping the sale and distribution as per Section 14 of the Drug Act 2035. Also, all concerned are requested not to recommend, sell, distribute and even use the drug.
Name of Medicine:
- LEVOSAFE-500mg (Levofloxacin 500mg Tablet IP)
- ZEFIX 100 (Cefixime 100mg Dispersible Tablet IP)
Department of Drug Administration Drug Recall Detail Notice

















Discussion about this post