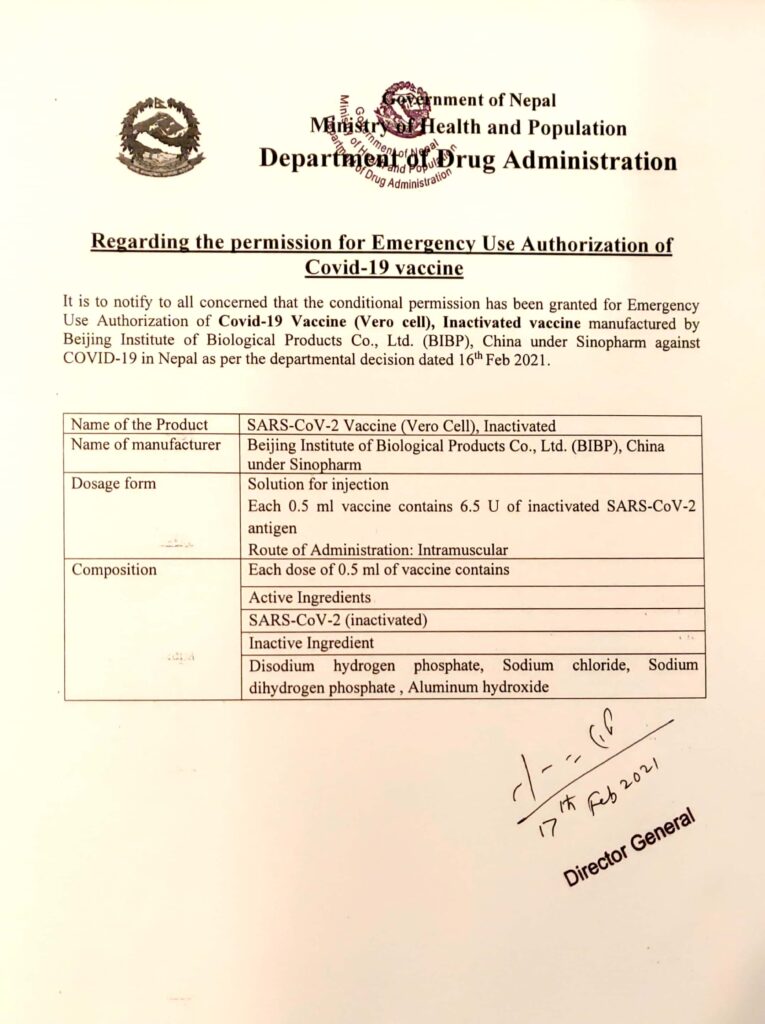
DDA has given Emergency use Authorization of Vero Cell vaccine in Nepal
EUA of covid 19 vaccine (Vero cell), inactivated manufactured by BIBP(under sinopharm)
DDA has given Emergency use Authorization of Vero Cell vaccine in Nepal
Department of Drug Administation – DDA notify that the conditional permission has been granted for emergency use of authorization of covid-19 vaccine (Vero Cell), Inactivated vaccine manufactured by Beijing Institute of Biological Products Co. Ltd. (BIBP), China under Sinopharm against COVID-19 in Nepal as per the departmental decision dated 16th feb 2021.
Name of Product: SARS-CoV-2 Vaccine (Vero Cell), Inactivated
Name of Manufacturer: Beijing Institute of Biological Product Co. Ltd. (BIBP), China under Sinopharm
Dosage form: Solution for injection. Each 0.5 ml vaccine contains 6.5 U of inactivated SARS-CoV-2 antigen
Route of Administration: Intramuscular
Composition: Each dose of 0.5 ml of vaccine contains
Active ingredients
SARS-CoV-2 (inactivated)
Inactive Ingredients
Disodium hydrogen phosphate, sodium chloride, sodium dihydrogen phosphate, Aluminum hydroxide.

















Discussion about this post